Sun, Sand, and Damaged RNA
by
 Some of you might have noticed the irregularity of my Mind the Gap posts of late. But fear not, I’m back! I defended my PhD a couple of weeks ago after having only 3 weeks to finish writing the thesis, so things got a little crazy. Crazy awesome: I’m now Katie PhD, for real!
Some of you might have noticed the irregularity of my Mind the Gap posts of late. But fear not, I’m back! I defended my PhD a couple of weeks ago after having only 3 weeks to finish writing the thesis, so things got a little crazy. Crazy awesome: I’m now Katie PhD, for real!
One of the bonuses of defending in the summer is that a lot of the ensuing celebrations took place outside in the sun, which for a Brit like me also meant several rounds of sunburn. Fortunately I keep a stash of aloe in the fridge for just such an occurrence to calm the red and the itchy, and I promise I wore sunscreen.
So it was with a two-day-old sunburn that I was cruising the interwebs looking for a paper to blog when I stumbled upon a study that implicated damaged non-coding RNAs in triggering the immune response to UV-damage. Having spent the last few years working on non-coding RNAs, I was (nerd alert) pretty excited, and printed myself a copy of the Nature Medicine paper.
Disclaimer: several mice were sunburnt in the making of this paper.
While it’s been known for some time that sunburn is an immune response to UV irradiation, the molecular trigger for this response has been a bit of a mystery. To address this question, Bernard and colleagues began by using a cell culture system that, like the cells in our bodies, responds to UV irradiation by producing several cytokines. They found that lysates from these irradiated cells could trigger a similar response in non-irradiated cells, but that this effect was lost if the lysates were treated with RNase. Injecting similar lysates into the ears of living mice, with “sunburn” only observed if RNA was present, further corroborated the idea that it was UV-damaged RNA that was causing the immune response.
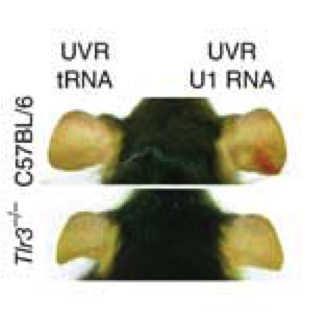
Mouse ears with U1-induced sunburn
Next, using a high throughput RNA sequencing approach, they found that there were changes in the structure of particular regions of snRNAs (non-coding RNAs involved in several nuclear processes, including mRNA splicing), and that these changes were likely candidates to activate ______ and produce the observed cytokine release. Sure enough, injection of UV-irradiated U1 snRNA induced “sunburn” in wild-type mice, but in ______ knockout mice the researchers saw no response.
These data open up a number of avenues for downstream applications, including the use of damage non-coding RNA as a biomarker for UV-induced damage and the development of alternatives to the phototherapeutic treatments used in several skin disorders.
Unfortunately, it would appear that the authors have no immediate plans to develop a super-sunscreen or anti-sunburn drug. I guess my fair-skinned compatriots and I will just have to stick to the old sunscreen and aloe combination for now.
.
Katie Pratt is a postdoc in Molecular Biology at Brown University. She has a passion for science communication, and in an attempt to bring hardcore biology and medicine to everyone, she blogs jargon-free at www.katiephd.com. Follow her escapades in the lab and online on Twitter.
.
Be the first one to mind the gap by filling in the missing gene as a comment and get your name in the blog along with a sweet new BenchFly mug!
.
UPDATE: Congratulations to Bryan Glezerson – this week’s Mind the Gap winner!
About the winner: Bryan is a researcher at the University of Calgary studying the microbiology of the cystic fibrosis lung. He’s particularly interested in mechanisms of Pseudomonas aeruginosa strain competition and “superinfection”.
About the prize: In addition to fame and glory beyond their wildest dreams, winners receive our new hot-off-the-presses large (15 oz) BenchFly mug to help quench their unending thirst for scientific knowledge… or coffee. Check out where the mug has traveled – will you be the first in your state or country to win one?
Miss a previous edition of Mind the Gap? Shame on you! Don’t worry – we’ve got you covered:
Let’s Talk About (Fruit Fly) Sex
This is Not an Article About Running Viruses
How is a Dormouse Like a Career Scientist?
With Great Power Comes Great Violin Strings
All the Better to See Sperm Whales With, My Dear
Saw VII: The Revenge of the Sawfish
Caution: Objects May Appear Larger Than They Really Are
Facebook Updates: The Good, The Bad, and The Vague
Scared of Dropping the Soap? Worry No More.
.



Bryan Glezerson
wrote on July 12, 2012 at 2:48 pm
TLR3!!!!!!!!
alan@benchfly
wrote on July 12, 2012 at 2:56 pm
We've got a winner!
Bryan Glezerson
wrote on July 12, 2012 at 3:03 pm
AWESOME!!! (great paper, btw…)
Charanya
wrote on July 12, 2012 at 4:01 pm
Tlr3